If you have been tracking the advanced therapies landscape recently, it has been hard to miss the growing momentum behind in vivo CAR‑T. What was once considered a longer-term scientific ambition is quickly emerging as one of the most active areas of investment and deal-making across cell and gene therapy.
The number of billion-dollar deals in this niche segment of life sciences is simply impossible to ignore. Eli Lilly, AbbVie, AstraZeneca, and Kite Pharma have all acquired in vivo CAR-T assets, whilst Strand Therapeutics, GRIT Biotechnology, Oricell Therapeutics, and Azalea Therapeutics have closed different funding rounds ranging from $65M to $153M. Taken together, these moves point to a market that is transitioning from early exploration toward structured pipeline development.
For service and solution providers supporting cell and gene therapy innovation, this shift marks a pivotal moment. As developer pipelines form and investment accelerates, demand for enabling technologies, platforms, and expertise is beginning to take shape in parallel.
The Shift is Already Underway
Across recent industry forums, including CAR-TCR Europe, conversations are increasingly shifting away from ex vivo approaches toward in vivo approaches as developers reassess scalability, manufacturing complexity, and long-term commercial feasibility. At the same time, capital is flowing into programs designed to move beyond bespoke, labor-intensive models toward more broadly applicable therapeutic platforms.
The "Gold Rush" Moment
A Head of Technology Development from Millipore Sigma recently described in vivo cell therapy as a “gold rush” saying “the gates have opened.”
In every gold rush, while product innovators capture headlines, some of the most durable value is often created by those enabling the market through infrastructure, tools, and specialized capabilities.
As in vivo programs progress, developers are increasingly assessing which partners can support delivery technologies, analytical development, manufacturing readiness, and regulatory alignment. For service providers, this stage offers an opportunity to embed early and shape long-term collaboration models before standards become fixed.
Capital Is Signaling Conviction
Billion-dollar deals are now shaping the in vivo cell engineering landscape, signaling serious intent from large pharmaceutical organizations that were previously cautious around cell and gene therapy modalities. This scale of investment reflects growing confidence in technical feasibility and a willingness to commit resources early.
This level of capital deployment from key players in the industry signals rapid expansion in pipelines and enabling infrastructure demand. For service providers, this is the time where positioning matters most.
Viral vs. Non-Viral: A Snapshot of Where the Field Is Heading
Capital allocation across delivery platforms offers a clear view of where the field is heading.
Viral vectors remain a clinically viable option and are often a natural starting point, supported by continued investment and partnerships. However, concerns around safety, immunogenicity, and regulatory complexity are contributing to growing interest in non‑viral approaches.
Non‑viral platforms, including lipid nanoparticles (LNPs) and mRNA-based systems, are attracting some of the largest recent deals in the space. Their potential for scalable manufacturing, repeat dosing, and broader application beyond oncology is driving developer interest and reshaping expectations around delivery solutions. For service providers, this shift highlights the importance of aligning capabilities with evolving platform preferences.
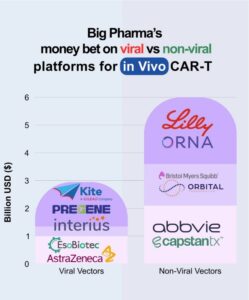
Source: Cesare Di Nitto – Crystal NAX
Why Positioning Ahead of Market Maturity Matters
Service and solution providers that engage with the in vivo ecosystem now stand to benefit in several important ways:
- Embedding their capabilities early on in platform and process development
- Building long-term partnerships as pipelines progress toward the clinic
- Establishing visibility and thought leadership while technical and regulatory standards are still being defined
- Avoiding entry into a later-stage market characterized by increased competition and pricing pressure
By contrast, waiting often means competing in a more crowded, price-driven market, once preferred partners and workflows are already defined.
A Narrow Window of Opportunity
The convergence of scientific readiness, positive industry sentiment, and accelerating capital deployment creates a limited window in which early engagement can still translate into category leadership. This moment will not remain open indefinitely.
For service and solution providers looking to position their organization at the center of this shift, the 5th In Vivo Cell Engineering & Gene Editing Summit offers a timely opportunity to engage directly with developers shaping the next generation of in vivo CAR‑T pipelines. As the field approaches its first major inflection point, those who contribute early will help define how this market evolves.
The gates are open. The only question is, who moves first?



